Fragrance Allergens: European Cosmetic Regulation Tightens

05 Mar 2026
Allergens: A core concern for the cosmetics and perfume industry
Fragrances are key components of cosmetics, but their ubiquity poses a major challenge: certain fragrance substances are common causes of skin allergies that affect a significant portion of the population. The European Union (EU) estimates that 1 to 9% of the population is allergic to fragrance substances, with allergic manifestations that require vigilance from manufacturers and clear information for consumers.
EU regulators are implementing multiple measures for both primary and secondary prevention. Primary prevention seeks to protect the population from developing fragrance allergies; secondary prevention aims to protect already‑sensitive individuals from experiencing allergic symptoms.
The European cosmetic regulatory framework challenged by allergens
Cosmetic products in the EU are strictly regulated by Regulation (EC) No 1223/2009, which imposes rigorous safety and labelling requirements. Recognizing the evolution of scientific knowledge on allergens, the EU later adopted Regulation (EU) 2023/1545.
The latter regulation amends EU cosmetic regulation Annex III to significantly strengthen consumer protection against allergenic fragrance substances, increase transparency, and expand the list of substances to be declared.
Allergens: What changes are expected in 2026?
Previously, Regulation (EC) No 1223/2009 required declaring 24 allergenic fragrance substances when their concentration exceeded a certain threshold.
Following Regulation (EU) 2023/1545, new restrictions are expected:
- Expansions to the allergen list: the list of individually declarable substances will more than triple—from 24 to 81 allergens—based on the opinion of the Scientific Committee on Consumer Safety.
- To simplify allergen labelling, certain substances will be grouped under a single designation called a “grouping name.” When determining if the labelling threshold is exceeded, one must consider the sum of all substances included under this designation, including all present sources, direct or indirect. If this regulatory threshold is exceeded, it is mandatory to use the relevant grouping name.
- Detailed regulatory thresholds for declaration: labelling will be mandatory when the concentration of allergens exceeds a certain regulatory threshold in the finished product:
- 001% for leave-on products (creams, perfumes, lotions, etc.); and
- 01 % for rinse‑off products (shampoos, shower gels, etc).
Transition periods and application deadlines: to allow industry to adapt, the following deadlines have been granted:
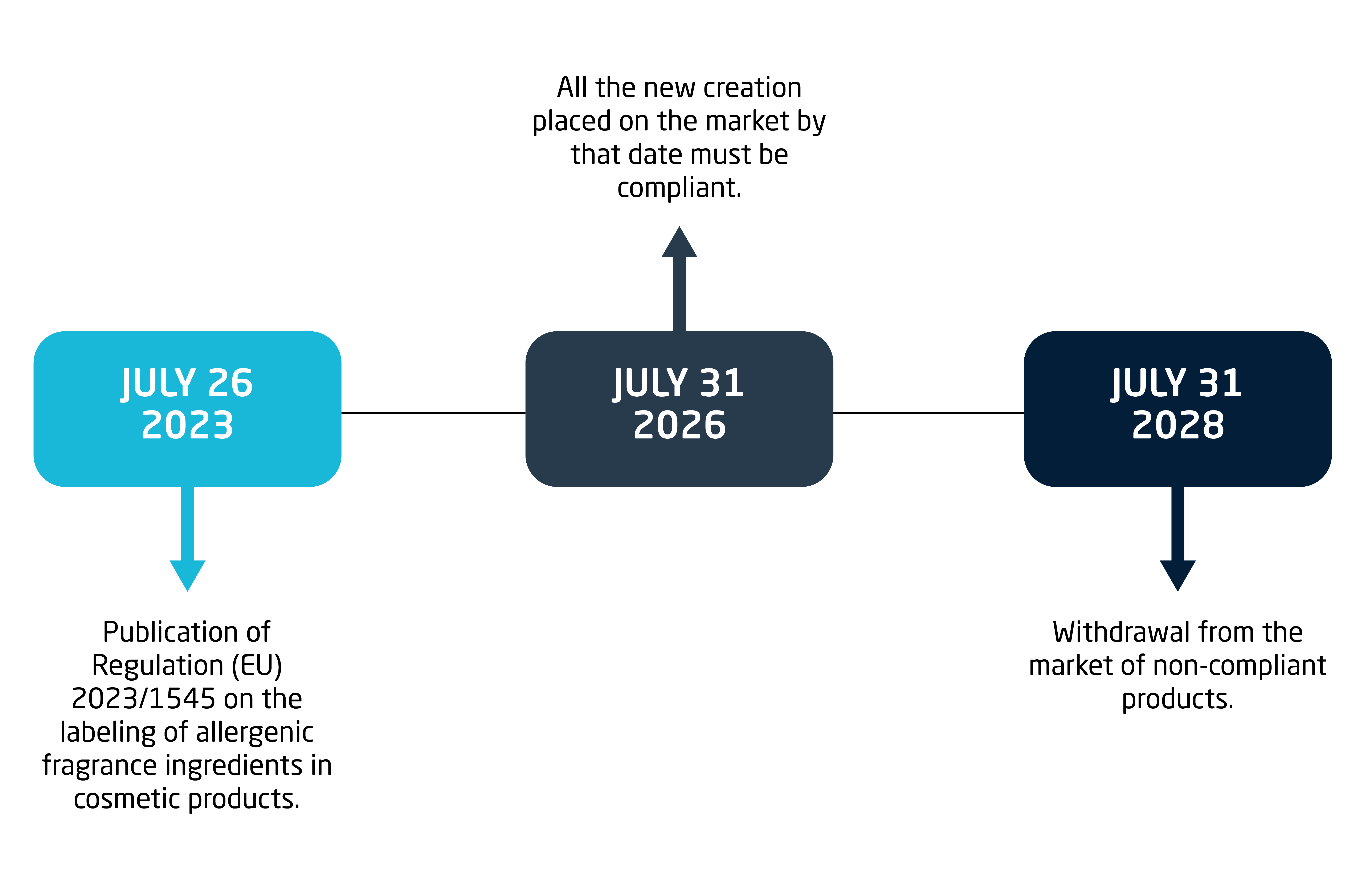
Conclusion: Towards safer and more transparent cosmetics
These new provisions represent a major advance in consumer safety, providing allergy-prone individuals crucial information to make informed choices. However, for industry, they may require thorough revisions of formulations, manufacturing processes, and labels. A rigorous understanding of this regulatory framework is essential to maintain product conformity and strengthen consumer trust in a constantly evolving sector.

